What is the difference between nuclear reaction and chemical reaction?
Students pursuing a chemistry course tend to learn different types of reactions where the nuclear and chemical reaction is no exemption.
The core difference between nuclear reaction and chemical reaction pdf is that nuclear reaction occurs inside the nucleus while chemical reaction occurs outside the nucleus.
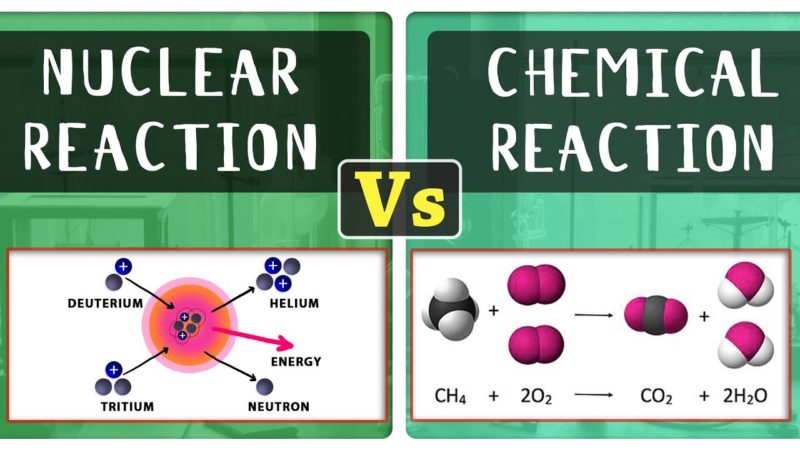
What Is Nuclear Reaction?
Nuclear reaction refers to a reaction where the nucleus of an atom collides with subatomic particles such as protons, neutrons, and electrons.
The reaction of these nuclides results in a different atom at the end of the reaction. The nuclear reaction causes heavy atoms to split into smaller atoms and even heavier form.
The formation of nuclear weapons and nuclear fission involves the use of nuclear reactions. This reaction occurs mostly in unstable and radioactive elements.
What Is Chemical Reaction?
Chemical reaction refers to the reaction that entails outermost electrons in an atom. The reaction results in the formation of a bond between the elements.
The atoms reacting are known as reactants and the result is known as the products. The type of bond occurring can either be an ionic or covalent bond.
The chemical reaction can move in a forward or backward direction until the equilibrium state is attained.
Some reactions can be spontaneous which implies that they do not require heat energy whereas others are non-spontaneous.
Comparison Chart: Nuclear Reaction Vs Chemical Reaction
| Basic Terms | Nuclear Reaction | Chemical Reaction |
| Meaning | It is the type of reaction that occurs in the nuclei of an atom | It is a type of reaction that involve the outer shell electrons in an atom |
| Elemental Composition | Tend to change due to the formation of new nuclides | Tend to remain the same but the bonding change |
| Energy Transformation | Massive amount of energy is released | Reactants need sufficient amount of energy for the reaction to happen |
| Occurrence | Highly unstable and radioactive atoms | The base of life and occurs at a given time |
| Rate of Reaction | Spontaneous | Non-spontaneous |
| Reactants | Atoms, ions, molecules, and compounds | Protons and neutrons |
| Nature | Decomposition of nucleus | Involve transfer, loss, gain, and sharing of electrons |
| Reversible or Irreversible | Tend to be irreversible | Can either be reversible or irreversible |
| Bond Formation | Nothing like that happens | Formation of new bonds after breaking the old bonds |
| Determinants | Independent reaction | Influence by pressure and temperature |
Core Difference Between Nuclear Reaction and Chemical Reaction
- A nuclear reaction is a type of reaction that occurs in the nuclei of an atom whereas chemical reaction is a reaction that entails outer shell electrons of an atom.
- The elemental composition of atoms involved in nuclear reaction tend to change while that of chemical reaction remains the same
- Nuclear reaction result in massive energy release while chemical reaction reactants need sufficient energy to take place
- The nuclear reaction occurs in highly unstable and radioactive atoms while chemical reaction can occur at any time in life
- The rate of reaction in a nuclear reaction is spontaneous while in the chemical reaction is non-spontaneous
- A chemical reaction is either reversible or irreversible while the nuclear reaction is irreversible
- Chemical reaction entails the breaking of old bonds and result in the formation of new bonds while nothing like that happens in a nuclear reaction
- The chemical reaction is influenced by pressure and temperature while the nuclear reaction is independent
- The reactants of a chemical reaction are atoms, ions, molecules, and compounds while nuclear reaction are protons and neutrons
- A chemical reaction involves the transfer, loss, gain, and sharing of electrons while nuclear reaction involves decomposition of the nucleus.
Read More: Difference between Entropy and Enthalpy
Comparison Video
Summary
The core similarities between nuclear reaction and chemical reaction are that both occur in an atom but the region of occurrence is what marks the difference. The ordinary chemical reaction is quite different from various types of nuclear reactions.